This section examines ways in which people all over the world are working to make diesel engines more socially useful and less harmful to the environment. The intensity of the effort is reminiscent of the 1960s and '70s when some of the best minds dropped out of college to live on pizza and play with computers. Many of these enthusiasts work within the confines of corporations, firms such as Bosch, Denso, Stanadyne, Magneti Marelli. Others pursue their dreams on their own. Go by a London fish-and-chips joint early in the morning and, chances are, you will find one of these pioneers collecting waste cooking oil to use as diesel fuel.
Brazil
In the summer of 2006, a group of students and technicians arrived at Vila Soledade , a remote village built along the banks of the Amazon. Like hundreds of other communities in the region, Vila Soledade is remote from the national grid and must generate its own electrical power. In theory, the 700 or so townspeople could have electricity for five or six hours a day. But diesel fuel is an expensive luxury for farmers living barely above the subsistence level and their antique gen-set frequently broke down. Sometimes they waited weeks for parts. In terms of the government's Human Development Index, the people in Vila Soledade are among the most disadvantaged in the country.
The team brought with them a Brazilian-built MWD TD229EC-6 generator powered by six-cylinder, turbocharged engine, modified to run on unprocessed palm oil. This oil is local product that costs nothing except for the labor involved in its extraction.
The modified engine circulates coolant through a holding tank to preheat the oil to 65 deg. C ( FIG. 1). A transfer pump then moves the warm oil through a filter to a second tank where it is heated to 85 deg. C for injection. With heat, the palm oil moves from the viscous esterarine phase to the more pumpable oleine phase. Sensors report fuel pressure and ambient, fuel, and exhaust temperatures, which are recorded daily by the operator.
Vila Soledade now has electricity for six hours every day. To prolong filter life the engine is run for a half hour on diesel fuel immediately after starting and just before shut down. The rest of the time it operates on raw palm oil.
Stack emissions on diesel fuel and palm oil were found to be almost identical, except that the vegetable oil produced no sulfur oxide. An analysis of the lube oil allowed change periods to be extended to 200 hours, which was surprising, since straight vegetable oil (SVO) has a reputation for contaminating lube oil. Misfiring developed after 600 hours due to dirty injectors. The injectors were changed and at 800 hours the head was lifted to decarbonize the combustion chambers. The major problem was fuel filters that gummed over and required frequent changes.
Reliable and affordable electricity has transformed the community. With the money saved on fuel, the villagers purchased equipment to process acaí, one of the major crops of the region, and many families have purchased lamps, television sets, refrigerators, and freezers. Most adults now attend night school.
While the Vila Soledade project impacts less than a 1000 people, it is part of a national commitment to wean the nation from fossil fuels. According to Luis Inácio Lula de Silva, the President of Brazil, "In the next 10 to 15 years, Brazil will become the most important country concerning renewable energies. . . . No one will have the ability to compete with us."
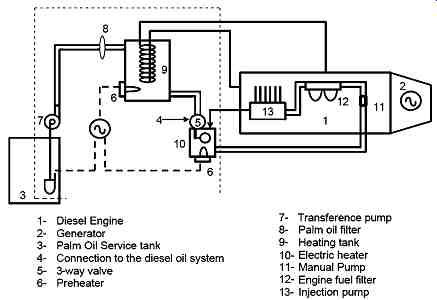
FIG. 1 SVO conversion kit developed by Brazilian government.
Lula's confidence seems well placed. According to the government, 43.8% of Brazil's current energy needs are met by renewable fuels, compared to 13.8% for the rest of the world. Ethanol has cut gasoline consumption almost in half, and a major push is underway to do something similar for diesel fuel.
The state oil company Petrobras squeezes as much as 20% more diesel fuel from conventional petroleum stocks by blending lighter hydrocarbons into the mix during refining. The tradeoff is a small reduction in heat value and a slightly lower flash point. Even so, the country still imports 10% of the diesel fuel it uses, which is a major draw on hard currency.
At the new century dawned, the government acted to reduce imports and, hope fully, to improve conditions in the agricultural sector. In only 12 months it set the stage for a massive influx of biodiesel by organizing production, establishing a regulatory framework, and arranging lines of credit.
In 2003, Petrobras introduced B2, a 2% blend of various vegetable oils and conventional diesel into most areas of the country and, by 2010, B5 will be universal. The program follows traditional practice, in that the raw vegetable oil undergoes transesterfication (reaction with methanol and caustic soda) for conversion to a fatty acid methyl ester, or FAME. This process reduces viscosity to something over 4 mm^2/s, which is in line with the European standard of 3.50-5.00 mm62/s. B2 has a density of 879 kg/m^3 compared with 860-900 kg/m^3 for European diesel.
B2 will yield an annual saving of 800 million liters of fuel and US $160 million that would be spent on imports. Several government agencies are working with Peugeot, International, and various parts suppliers to make B100 practical for agricultural machinery and portable power plants. Locomotives will, it is predicted, soon operate on B25 and, with some modification, heavy trucks should be able to use B10.
In addition, the Brazilians hope to export FAME to Europe, where legislation mandates 5.75% renewables by 2010. Europe does not have the agricultural base to support such a commitment.
In a parallel program, Petrobras has converted three refineries and is building several more to process vegetable oil by hydrogenation. According to the company, H-bio blends have superior ignition characteristics and have shown themselves to be harmless to fuel systems.
Researchers at the University of Brasilia have explored pyrolysis as a means of converting soybean, palm, and castor oils to diesel-compatible fuel. 2 The technology is not new: pyrolysis was widely used in China during the Second World War to convert ming oil into feed stocks, which were then used to produce gasoline and diesel.
The university researchers used a stainless-steel reactor to distill vegetable oil into four fractions, at 80 deg. C, 80 deg. -140 deg. C, 140 deg. - 200 deg. C, and >200 deg. C. These moderate temperatures left a heavy residue accounting for about 2% wt of the oil in the apparatus, which was discarded. As shown in Table 1, the distilled fractions have all of the salient characteristics of diesel fuel.
U.S. and Europe
Mixtures of FAME (vegetable oils converted to fatty acid methyl esters) and conventional diesel fuel are a fact of life in Europe and the United States. B2 is widely available in this country and provides the assurance of adequate lubrication that may be needed with low-sulfur (15 ppm) fuel. Europe is moving to a B5 standard and may adopt a B20 standard in the near future.
Renewable fuels appeal to something deep in human nature and, unlike petroleum fuels, do not contribute to global warming. Rather than collecting in the atmosphere, the CO2 released by burning vegetable products is recycled back into plant growth.
A joint statement issued by Denso, Bosch, Dephi, Siemens VDO, and Stanadyne approves the use of B5 if, the FAME content conforms to the EURO EN 14214 standard and the base petroleum fuel to EURO EN590. In contrast to ASTM (The American Society for Testing Materials) standards for fuels sold in this country, EURO standards include resistance to oxidation. FAME is biodegradable, which means that it oxidizes rapidly in the presence of heat, water, and various metallic ions. By products of oxidation include solids and highly corrosive formic and acetic acids.
Other potential problems that fuel-system makers identify with FAME are listed in Table 2.
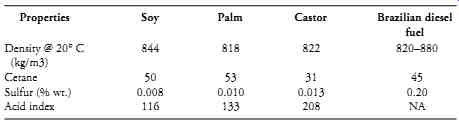
Table 1. Physical and chemical characteristics of distilled vegetable oil
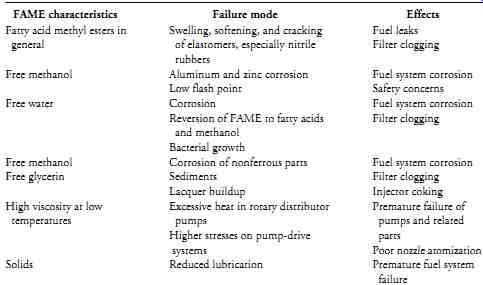
Table 2. Possible effects of FAME on fuel systems
===
FAME characteristics Failure mode Effects Fatty acid methyl esters in Swelling, softening, and cracking Fuel leaks general of elastomers, especially nitrile Filter clogging rubbers Free methanol Aluminum and zinc corrosion Fuel system corrosion Low flash point Safety concerns Free water Corrosion Fuel system corrosion Reversion of FAME to fatty acids Filter clogging and methanol Bacterial growth Free methanol Corrosion of nonferrous parts Fuel system corrosion Free glycerin Sediments Filter clogging Lacquer buildup Injector coking High viscosity at low Excessive heat in rotary distributor Premature failure of temperatures pumps; pumps and related Higher stresses on pump-drive parts systems Poor nozzle atomization Solids Reduced lubrication Premature fuel system failure
===
Consequently, the manufacturers say they "accept no legal liability attributable to operating their produces with fuels for which their products were not designed, and no warranties or representations are made as to the possible effects of running their produces with such fuels." In other words, American B2 users are on their own, without warranty protection.
Yet many of the same corporations produce fuel-system components used with Brazilian B2 that soon will be replaced with fuels of higher FAME percentages.
According to the government, all fuel-system warranties will be honored. Brazil does not ascribe to the International Monetary Fund/World Bank model of development and, consequently, has a great deal of leverage over multinational corporations doing business in the country.
DIY FAME
Several companies market DIY transesterfication plants to convert vegetable oil into fatty acid methyl ester. There is no guarantee that the resulting FAME conforms to the ASTM D6751 standard for the commercial product, but the critical parameter-viscosity-is reduced and most solids and water are eliminated. By using waste cooking oil as the feedstock, the cost of the fuel comes out to less than $1 a gallon.
Restaurants usually give the oil away, since they would otherwise must pay to have it removed. Nor is there any shortage of the product: according to one estimate, three billion gallons of cooking oil are discarded every year in the United States.
Extreme Biodiesel, based in Orange, California, markets the Eliminator, consisting of two large plastic tanks, pumps, filters, and associated plumbing. The apparatus can be set up in a garage and will, its makers say, produce 100 gal of diesel fuel in 12 hours.
The process begins with pumping the oil through a primary stage of filtration to remove large solids. This is followed by titration, a simple chemical test to determine the amount of methanol and caustic required for conversion. The user then mixes the chemicals with the oil, which is heated to speed the process. Several hours later, glycerin, the by-product of transesterfication, drops out.
Impurities that remain are removed by water spray, passed through a water separator and a 15 _m filter. An alternative to the water spray is to treat the fuel with Magnesol, a decontaminate widely used in the food processing industry. At this point, the FAME is ready to be mixed with conventional diesel fuel.
Straight vegetable oil
It is possible to operate diesel engines on straight vegetable oil (SVO) on the model of the Vila Soledade palm-oil project. Rudolf Diesel's first commercial engines, one of which was demonstrated at the 1900 Paris World Fair, ran on straight peanut oil. The inventor, who had no love for oil companies, wanted farmers to be able to produce their own fuel. Even after it became clear that SVO could not compete with cheap petroleum, Diesel struggled for years to adapt his engines to run on coal dust.
Rapid increases in oil prices during the 1970s sparked a new interest in SVOs in Europe. A leading figure in this movement was Ludvid Elsbett (1913-2003), an engineer of remarkable talent who held more than 400 patents. In 1937 he was hired as a department manager for Junkers Motorenwerke AG, with responsibilities for the gasoline direct injection system used in the Jumo 211 engine that powered the JU 87, JU-88, and other military aircrafts. As a point of interest, the DI system consisted of 1575 individual parts, many of them precision lapped. 3 After the war he was employed by the truck manufacturer MAN and, during the 1970s led an abortive attempt to adapt alternative fuels to diesels in Brazil.
Subsequently, Elsbett and his sons opened a shop in Bavaria that became the first, if not the only, commercial source of SVO automobile engines.
Thousands of enthusiasts, working from information on the Internet, have converted their cars and light trucks to burn SVO. The type of oil used is determined by price and availability. Rape-seed oil is favored in Europe and sunflower-seed oil in this country. Many of these enthusiasts run their cars on waste cooking oil, unprocessed except for filtration.
But SVO is not without problems. One of the most intractable is the way vegetable oil carbons over injectors and combustion chambers. FIG. 2 graphs the coking effects of sunflower oil against No. 2 diesel. Note that carbon deposits increase almost linearly with the amount of SVO mixed with diesel. This imposes a fairly severe maintenance requirement and, even with frequent de-carbonization, does not obviate the possibility of reduced engine life due to skewed nozzle spray patterns and contaminated lube oil.

FIG. 2 Coking, that is, how quickly carbon is deposited on internal engine
parts, increases with the percentage of SVO added to conventional fuel.
Straight vegetable oil also presents a viscosity problem at low temperatures. As shown in FIG. 3 sunflower oil is an order of magnitude more viscous than No. 2 diesel at 18 deg. C. The high viscosity of SVO is enough to make it an outlaw fuel, unrecognized by diesel manufacturers and government agencies. People who use the fuel mitigate the viscosity with heat and by mixing the SVO with diesel, alcohol, and even gasoline. SVO also attacks seals, hoses, and other elastomer parts.
The best candidates for conversion are older, IDI engines with Bosch PE-type inline pumps and pintle injectors. The pumps are nearly indestructible and the injectors can be easily disassembled for cleaning. Distributor-type injector pumps should be avoided, since lubrication is entirely fuel dependent and electronic versions of these pumps cut off fuel delivery during coastdown. There have been reports that the alcohol present in FAME de-laminates the internal timing sensor (the sensor that tracks pump-cam position relative to the engine camshaft) on Bosch PV44 radial piston pumps.
Most SVO conversions use conventional diesel fuel for starting and have a second tank, heated by engine coolant, for vegetable oil. A thermostatically con trolled resistance heater at the suction side of injector pump takes some of the strain off the pump. Manually controlled glow plugs can remain energized until engine temperatures stabilize.

FIG. 3 At room temperature, SVO, in this case sunflower-seed oil, has an
order of magnitude greater viscosity than No. 2 diesel.
Dual-fuel engines
With some tradeoff in performance, diesel engines can be made to run on a mix ture of diesel fuel and compressed natural gas. Benefits are reduced NOx and PM emissions, lower fuel costs, and for the five Caterpillar 3170B trucks operated by Pima Gro Systems in California, full warranty coverage. The gas, injected into the manifold at 125 psi, is the primary fuel, ignited by small amounts of No. 2 diesel.
A dual-fuel ECU adjusts pulse width for both sets of injectors with respect to manifold pressure, charge-air and gas temperatures, and fuel mapping. At low rpm the engines "skip fire," with three cylinders receiving diesel fuel and no fuel to the others. As loads and speeds increase, progressively greater amounts of natural gas are injected and the remaining three cylinders come on line. Skip firing reduces emissions and provides the surplus air to ease the transition from one fuel to the other. These trucks run about 350 miles a day without major problems.
Conventional fuel
Serious efforts are underway to ameliorate some of the worst effects associated with conventional fuels. In accordance with the Kyoto Protocols, the EU has agreed to reduce emissions of CO2, which are believed to be the primary cause of global warming. Because diesels produce less carbon dioxide than SI engines, European governments promote their use with tax breaks and diesel-friendly emissions regulations ( Table 3). Many experts believe that, by 2008, half of the new cars registered in Western Europe will be diesel powered.
Some translation is needed:
• PM (particulate matter) refers to the solids in diesel exhaust that are some times visible as soot.
• NOx (nitrogen oxides) is a blanket term for various oxides of nitrogen, such as NO and NO2.
• HC (hydrocarbons) come about as the result of incomplete combustion of fuel and lube oil.
• CO (carbon monoxide) is an odorless gas, lethal at high concentrations.
Table 4 lists the U.S. Environmental Protection Agency (EPA) 50-state standards for light vehicles that were phased in during 2004 with full compliance scheduled for 2010. These standards apply both to diesel- and gasoline-fueled vehicles.
Heavy pickup trucks, such as the Silverado, and large SUVs are partially grandfathered until 2008, when they will have to meet the same standards as other vehicles.
What the EPA calls NMHC (non-methane hydrocarbons) is known as HC. HCHO, better known as formaldehyde, is a powerful carcinogen.
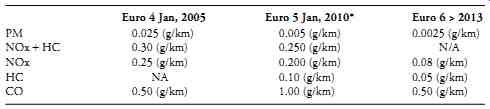
Table 3. Current and pending EURO exhaust emissions regulations
In the EPA scheme of things, manufacturers are free to classify their engines into any of the seven bins shown in the table. But the fleet average, that is, emissions from all engines the manufacturer sells, must conform to Bin 5 standards after 2010. Rather than juggle the mix by counter-balancing relatively dirty Bin 7 or 8 engines with super-clean Bin 2 or 3 models, most manufacturers aim at producing Bin 5 engines across the board.

Table 4. EPA Tier 2 light-duty vehicle emissions standards
NOx and PM
Bin 5 limits NOx to 0.07 g/mile and PM to 0.01 g/mile, which are, respectively, one-sixth and one-half of EURO 4 limits. The PM standard approaches that of rubber dust from vehicle tires. Some explanation is required to understand why the EPA has taken these draconian measures, which affect diesel engines far more than their SI counterparts.
Both NOx and PM pose serious health risks, especially for urban populations.
NOx reacts with sunlight to produce smog and is the pollutant most responsible for low-level ozone. The health effects of long-term exposure to ozone include chronic respiratory problems, reduced potential for exercise, and more frequent emergency room visits. Ironically some of the most severe ozone exposure occurs in suburbs, downwind of city traffic.
PM, sometimes visible as the sooty component of diesel exhaust and felt as a stinging sensation on the face, presents an array of health problems, one of which is lung cancer. According to the American Lung Association, 50,000 Americans die each year because of exposure to PM, taken for purposes of the study as particles of less than 10 _m in diameter. No one has identified the biological mechanism involved, nor does the association try to distinguish between diesel PM, dust, and tobacco smoke. But there is a strong statistical correlation between inhaling PM and dying early. Perhaps a more telling statistic is that between 1980 and 2000, asthma increased 160% in American children under the age of four. African-American children, who live in disproportionate numbers in urban areas with high levels of diesel PM, die from asthma at the rate 11.5 per million, or more than four times the rate for white children.
Diesel engines are relatively clean in terms of HC and CO, but produce large amounts of NOx and PM. By insisting that all engines, regardless of fuel used, meet the same emissions standards, the EPA has moved to close what it identifies as the "health gap" between CI and SI.
Unfortunately, the two most toxic pollutants in diesel exhaust are the most difficult to control. The same high combustion-chamber temperatures that burn off HC and convert CO to CO2 generate NOx and PM.
Designers were able to meet EPA Tier 1 and EURO 4 NOx and PM standards with in-cylinder controls, such as the use of exhaust gas recirculation and retarded injection to reduce flame temperatures. However, Tier 2 and pending European standards cannot be met by engine modifications alone. For that, one needs exhaust aftertreatment.
NOx aftertreatment
The high level of air in diesel exhaust makes the three-way catalytic converters used on SI automobiles impractical for CI engines. But it is possible to engineer around the problem, although at the price of some complexity.
FIG. 4 illustrates the system developed by Houston Industrial Silencing for standby generators and other stationary applications. It covers all bases with a highly efficient muffler, two catalytic converters and a PM trap. The microprocessor controlled injection system delivers the correct amount of ammonia (NH3), to the first-stage converter. Ammonia reacts with NOx to produce free nitrogen and water.
NH3 + NOx = N2 + H2O
Stated more precisely to account for the NO and NO2 components of NOx, the reaction goes.
4 NO + 4 NH3 + O2 = 4 N2 + 6 H2O 2 NO2 + 4 NH3 + O2 = 3 N3 + 6 H2O
A second converter, downstream of the first, causes HC and CO to react with surplus O2 in the exhaust to yield carbon dioxide and water:
HC + CO + O2 = CO2 + H2O
A solution of 75% water and 25% ammonia is normally used as the oxidant. The dry (anhydrous) form of ammonia can also be injected directly into the exhaust or mixed with steam prior to injection. In any event, the rate of injection must be precisely calibrated for each installation by sampling NOx concentrations at the exhaust outlet.
When using platinum as the catalyst, the system is said to reduce 90% or more of the NOx present and at least 95% of the CO. HC conversion rates are comparable to those for CO. Platinum operates in a temperature band of 460°-540°F, attainable by a well-engineered installation operating under constant load.
NOx-reduction systems for motor vehicles pose more formidable problems ( FIG. 5). These systems must be compact, capable of operating under variable loads and, when used on heavy trucks, must function 290,000 miles without maintenance. For automobiles, the zero-maintenance requirement is 100,000 miles.
Ford and Bosch employ a solution of urea (NH2)2CO and water as the ammonia carrier. Urea breaks down into ammonia (NH3) in the catalytic converter, which reacts with NOx as described above to produce water and nitrogen gas. Under ideal temperature conditions, these systems are claimed to oxidize more than 90% of avail able NOx. The urea component amounts to about 5% of diesel fuel consumption.

FIG. 4 A typical stationary engine installation that combines ammonia dosing
with a PM trap and efficient sound deadening.
A delivery system filters the urea, atomizes it with compressed air, and adjusts delivery to engine load, speed and exhaust temperature. One or more oxygen sensors continuously sample the exhaust to provide feedback.
AdBlue, the Bosch marketing name for urea (actually a common fertilizer), is available at service stations in Germany. Ford and Mercedes-Benz have received EPA approval to introduce these converters in the U.S. although the agency would prefer a true zero-maintenance system.
Honda's 2.2 L i-CTDi, scheduled for introduction before 2010 when Tier 2, Bin 5 comes into effect, does away with urea injection. The catalytic converter is coated with two absorbent layers. The outer layer traps NOx during lean-burn operation. At intervals, the engine goes rich. Hydrogen, obtained from the unburnt fuel in the exhaust, reacts with the captive NOx to produce ammonia (NH3). The second layer of absorbent material stores NH3 until the engine returns to its normal, lean-burn operation. At this point, the ammonia reacts with NOx to yield water and nitrogen gas.
The system should have very little effect upon fuel consumption: according to a Honda spokesman, when running at 60 mph the engine operates lean for three minutes, then rich for five seconds. Amazing.

FIG. 5 Bosch aftertreatment system for motor vehicles. The particulate trap
(7) converts NOx to NO2, which then oxidizes the carboniferous components of
soot into CO2. Those components that escape oxidation collect in the filter
element. A differential pressure sensor (3) monitors the pressure drop across
the trap. When backpressure becomes excessive, the ECU initiates regeneration
by briefly supplying more fuel than the engine can use. The surplus fuel ignites
in the particulate trap to burn off the soot and return backpressure to normal
levels.
Sensors (2 and 5) monitor exhaust temperatures going into and leaving the catalytic converter (6).
PM trap
The trap, also called a diesel particulate filter (DPF), mounts upstream of the catalytic converter. Because of the high temperatures involved, filter elements are made of porous ceramic or sintered metal. When the filter clogs, the computer senses the restriction as excessive backpressure, and orders injection late during the expansion stroke. Raw fuel floods the trap and spontaneously ignites, burning off carbon and soot accumulations. Periodically, the trap must be disassembled for removal of ash and other hard deposits.
Peugeot-Citroen employs a honeycomb silicon carbide PM trap that is cleaned with a combination of fuel and a proprietary additive, known as Eolys. Since Eolys is not the sort of stuff you buy at the corner gas station, the EPA will probably not approve it.
Ultra-low sulfur diesel Sulfur clogs PM traps and poisons catalytic converters, and is in itself a source of PM. Consequently, the EPA ruled that the sulfur content of Nos.1 and 2 diesel be reduced from 500 ppm to 15 ppm. ULSD (ultra-low sulfur diesel) may be used in any vehicle engine, and is mandatory for 2007 and later models.
The changeover, which begun in mid-2006, put at least one small refinery out of business, but has generally progressed smoothly. One concern was that hydrogenation, the process refiners use to remove sulfur, also lowers the lubricity of ULSD.
Additives bring the fuel up to 500 ppm lubricity levels. Aside from scattered reports, difficult to document, there seems to have been no serious problems in this regard.
Because diesel engines live so long, it will be decades before the full effect of Tier 2 standards and ultra-low sulfur fuel make themselves felt. If EPA projections are correct, our grandchildren should see a 2.6 million ton reduction in annual emissions of NOx and a 100,000 ton reduction in PM. Meanwhile, the regulatory climate in the U.S. is changing. The reality of global warming has now been accepted by most opinion leaders, including many of the same conservatives who led the fight against ratification of the Kyoto Accords.
Effects of other emissions are, for the most part, invisible and detected only by statistics on hospital admissions and longevity. Signs of catastrophic climate change are more difficult to ignore.
Environmentalists hailed the recent Supreme Court decision that empowers the EPA to treat carbon dioxide emissions under its general mandate to protect the environment. Other pollutants can be converted chemically into less noxious com pounds. But no conversion technology, no magic bullet, exists for CO2. The only way to reduce carbon dioxide emissions is to burn less fuel. As the most efficient form of internal combustion, the diesel engine has a bright future.
Prev. | Next
Home top
of page Similar articles